Gothenburg, August 16, 2025 — Novo Nordisk has just made strategic history: the U.S. Food and Drug Administration granted accelerated approval for its once-weekly GLP-1 therapy, Wegovy (semaglutide 2.4 mg), to treat metabolic dysfunction-associated steatohepatitis (MASH)—a serious form of fatty liver disease. It’s the first GLP-1 drug to earn this indication in the United States, and the move has already sparked a meaningful reaction in the markets.
Clinical Impact and Market Opportunity
The approval builds on strong results from Part 1 of the Phase III ESSENCE trial:
- 63 % of Wegovy-treated patients achieved resolution of steatohepatitis without worsening fibrosis—versus ~34 % on placebo.
- 37 % saw improvement in liver fibrosis without worsening inflammation—compared to ~22 % with placebo.
- About one in 20 U.S. adults have MASH—a sizeable enough pool to meaningfully expand Wegovy’s addressable market.
This fills a long-standing therapeutic gap. Until now, only Madrigal Pharmaceuticals’ Rezdiffra has an FDA-approved indication for MASH, which stacks Wegovy with a first-mover advantage.
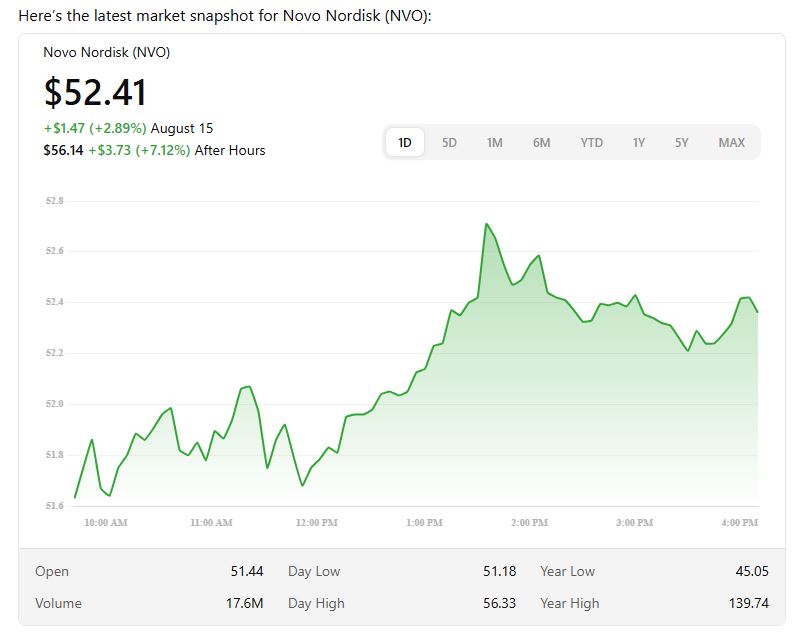
Market Reaction: Stock Moves After Hours
Investors didn’t wait for Monday. Novo Nordisk stock jumped ~7 % in after-hours trading following the FDA announcement. That kind of premium reflects the market recognizing the vast potential of this new indication.
Strategic Implications and Growth Trajectory
Let’s break down what this really means:
- Diversified revenue streams: Wegovy already carries weight as a blockbuster for obesity and recently for cardiovascular risk reduction. Now, MASH adds another high-growth corridor.
- Global rollout in sight: Regulatory submissions for MASH are already underway in the EU and Japan, with part 2 results expected by 2029.
- Strategic pricing and access: Novo is countering market headwinds from compounded knockoffs by lowering cash prices—Wegovy now can be accessed at ~$499/month through NovoCare, down from ~$1,350. This improves affordability and keeps patients on licensed product.
- Competition and the broader GLP-1 space: Eli Lilly’s tirzepatide (Zepbound) has shown promising MASH data in mid-stage trials. But Wegovy’s approval gives Novo a significant head start.
The Bottom Line
Novo Nordisk did two things today: it flipped a large unmet medical need into a commercial opportunity, and it validated its GLP-1 strategy yet again.
For Nordic investors: Wegovy’s MASH label broadens both the company’s therapeutic footprint and its revenue runway. With the U.S. move signalling potential regulatory momentum elsewhere, this isn’t a one-off. It’s a calculated expansion of the semaglutide franchise.
Expect more investor interest, especially if early trade continues to reflect this optimism.
