By Lina Halvorsen, Senior Correspondent, Copenhagen
On the 14th floor of Ferring Pharmaceuticals’ campus in Øresund, a 3-D hologram is looping on a 4-metre screen. It shows a translucent, six-day-old human embryo sinking—almost melting—into a synthetic uterine wall. Each twitch of its cells is being watched by strategists, patent lawyers and IVF-clinic owners flown in from Oslo, Helsinki and Stockholm for an invitation-only briefing.
The clip is only 18 seconds long, but according to McKinsey’s latest Nordic life-science outlook it could unlock an €11 billion regional market over the next decade.
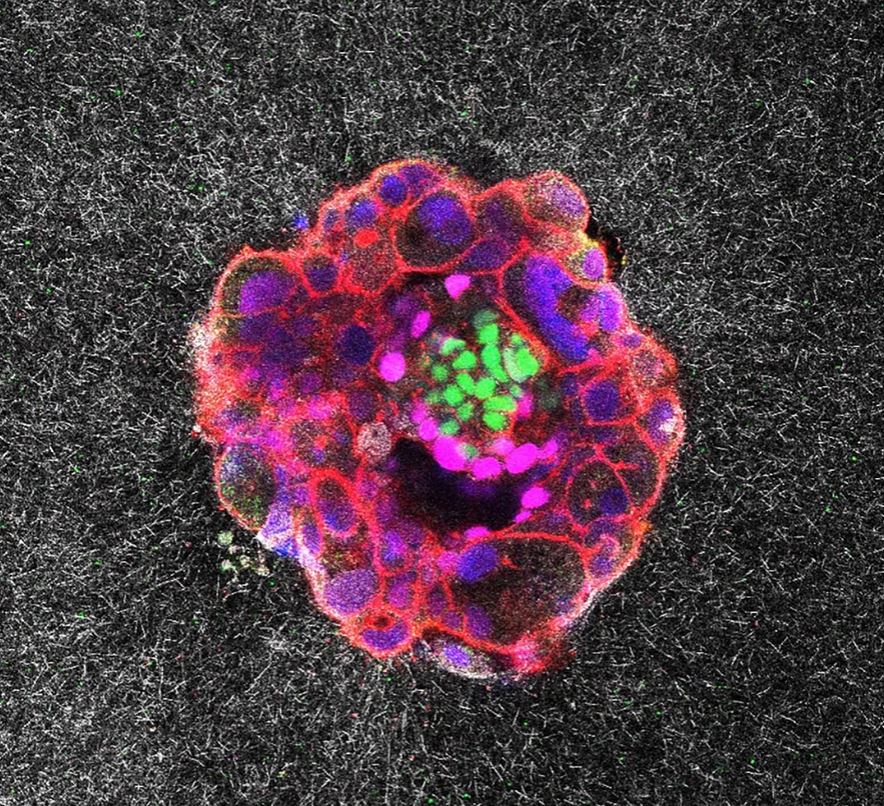
The film is the world’s first real-time footage of human implantation, published last week in Science Advances by a Barcelona team led by Dr Samuel Ojosnegros. Behind the Spanish headlines, Nordic players have already staked claims in the intellectual-property pipeline. Here’s why the footage matters to balance sheets north of the 60th parallel.
A New Diagnostic Moat
Today, 60 % of IVF failures happen at implantation—a €3.2 billion Nordic IVF market that has plateaued on 35 % live-birth rates. The Barcelona group mapped traction-force signatures that distinguish embryos likely to implant from those that will not.
- Kaivogen Oy (Espoo) filed a provisional patent last Wednesday to license the force-mapping algorithm for its time-lapse incubators.
- Origio A/S (Malmö), acquired by CooperSurgical in 2022, is negotiating an exclusive Nordic distribution agreement.
“For clinics, the diagnostic adds maybe €300 to a €4,000 cycle,” says Kaivogen CEO Dr Aino Virtanen. “But if we lift success rates to 50 %, patients will pay.”
Synthetic Uterus Models Replace Animal Trials
EU regulation is phasing out animal testing for reproductive drugs by 2035. The Barcelona team’s collagen-protein matrix offers an alternative that regulators already classify as “non-animal, human-relevant.”
- Novo Nordisk’s Bio Innovation Hub in Måløv is co-funding a €12 million scale-up to turn the matrix into a 96-well screening platform.
- Biosyntia (Copenhagen) is prototyping a sustainable, plant-based collagen supply chain to cut material costs by 70 %.
Pharma: From Fertility to Oncology
The same force-mapping technology can be repurposed to study how tumours invade tissue. Ferring has quietly moved its R&D chief, Dr Kristina Åkerberg, to a stealth programme on endometrial cancer metastasis, using the implantation model as an in-vitro invasion assay.
Barcelona’s IP is under a Creative Commons licence for academic use, but commercial rights are administered by IBEC’s tech-transfer office. Nordic legal firms AWA and Zacco have already filed 14 PCT applications on downstream uses.
The Data Play
Every microscope in the project streams to Amazon Web Services’ Stockholm region. The raw dataset is 2.1 petabytes; anonymised force-maps are being sold to AI companies training digital-twin embryos.
- Sana Labs (Stockholm) signed a €2 million data-licence deal last month.
- Novo Holdings’ RE:Invest has earmarked DKK 50 million for Nordic start-ups that can turn the data into predictive SaaS tools for clinics.
Regulatory Fast-Track
The footage arrives as the European Medicines Agency finalises its Guideline on Implantation-Related Medicinal Products, due Q2 2026. Nordic regulators—traditionally pragmatic on reproductive tech—are expected to pilot an “implantation passport” allowing simultaneous approval in Sweden, Denmark and Finland.
The Nordic Scorecard
| Company / Institution | Angle | Capital Raised since 1 Jan 2025 | Next Milestone |
|---|---|---|---|
| Kaivogen Oy | Force-mapping diagnostics | €8 m Series A | CE-mark by Q4 2026 |
| Origio A/S | Distribution rights | — | Nordic tender Q1 2026 |
| Novo Nordisk | Oncology assay | €12 m internal | GLP validation Q2 2026 |
| Ferring | Endometrial-cancer pipeline | Undisclosed | IND filing 2027 |
| Biosyntia | Sustainable matrix | €5 m seed extension | Pilot plant Q3 2026 |
Voices from the Front Line
Dr Samuel Ojosnegros, IBEC:
“We chose Nordic partners because they move fast on translational research. The regulatory predictability here is better than in the U.S.”
Ellen Trane Nørby, Chair, Danish Parliament’s Health Committee:
“This technology aligns with our national strategy to become Europe’s preferred test-bed for reproductive innovation.”
Pia Höglund, patient advocate, Stockholm:
“If clinics can finally tell me why my embryo didn’t stick, I’ll pay out-of-pocket. The emotional ROI is enormous.”
Risk Radar
- Ethics backlash: Norwegian Biotechnology Advisory Board is reviewing whether synthetic-uterus models circumvent the country’s strict embryo-research laws.
- Market saturation: If every Nordic clinic adopts the diagnostic, price erosion could start by 2028.
- Data privacy: Swedish Authority for Privacy Protection (IMY) is auditing cross-border embryo-data transfers to AWS.
Bottom Line
For a region already holding three of the world’s top five IVF success rates, the footage is more than a scientific milestone. It is the rare innovation that shortens the gap between bench and bedside to roughly 36 months.
As the hologram ends its loop in Ferring’s boardroom, the CFO clicks to a slide titled “Revenue Scenarios”. The most bullish line—assuming Nordic clinics adopt the diagnostic at 80 % penetration—shows an EBIT uplift of €1.6 billion by 2030.
In the corridor outside, a venture partner from EQT Life Sciences is already dialling Barcelona. The race for the €11 billion implantation economy has begun—and, for once, the starting gun was fired in the Nordics.
