As more Danes continue to pour investments into Novo Nordisk, the Danish pharmaceutical powerhouse is ramping up its legal fight against counterfeit versions of its blockbuster drugs in the United States. On August 5, Novo Nordisk announced it has filed 14 new lawsuits against companies selling unauthorized, often unsafe, versions of semaglutide—the active ingredient in its highly sought-after diabetes and weight-loss drugs Ozempic and Wegovy.
These lawsuits target both compounding pharmacies and telehealth companies that, according to Novo Nordisk, steer patients toward illegal knockoff products under the pretence of personalized medicine. The Danish firm alleges that these entities violate regulations by misleading patients and doctors, falsely suggesting that their compounded products have FDA approval or equivalent effectiveness and safety to Novo Nordisk’s own.
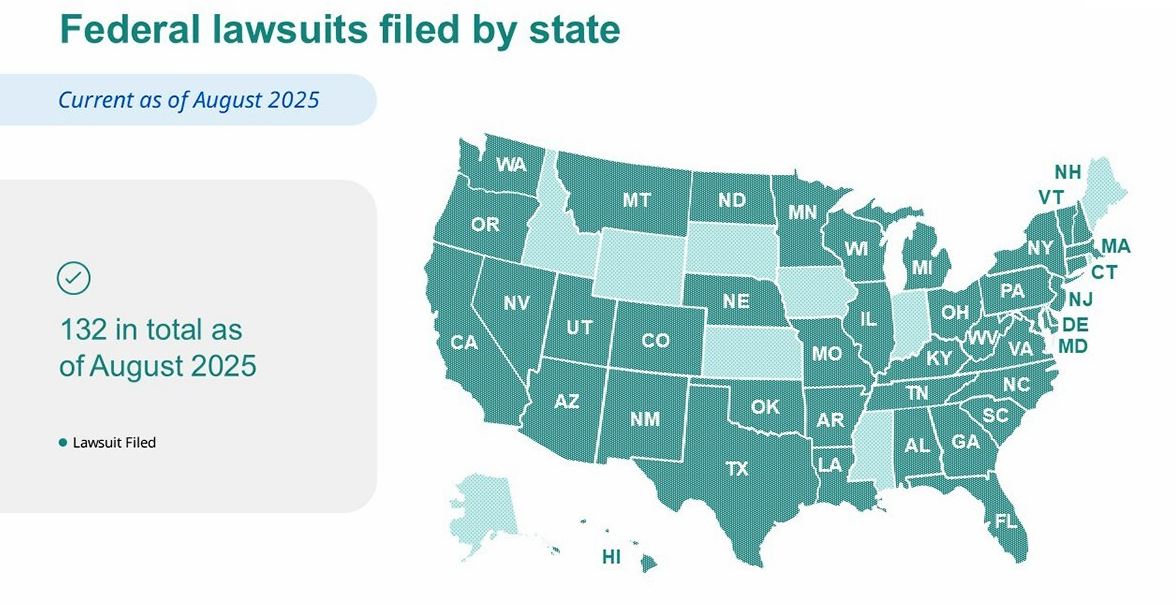
These legal steps add to an already extensive campaign. Novo Nordisk has now initiated more than 130 lawsuits across 40 states, with U.S. courts having issued at least 44 permanent injunctions prohibiting the marketing and sale of these counterfeit drugs. Some of these knockoff medications pose significant risks, ranging from mislabelled strengths to high levels of chemical impurities.
The U.S. Food and Drug Administration (FDA) has issued multiple warnings on the issue, alerting healthcare providers and the public to the dangers of counterfeit Ozempic found in the legitimate supply chain. In April 2025, the agency seized hundreds of counterfeit units and advised pharmacies and patients to be vigilant, reporting any suspicious products directly to federal authorities.
Despite intense legal and regulatory scrutiny in America, Novo Nordisk’s popularity among Danish investors has remained strong, even after recent market turbulence. Confidence in the company’s global prospects and leadership in the new obesity treatment market continues to outweigh concerns over temporary revenue or litigation setbacks.
Novo Nordisk’s legal campaign sends a clear message: the global pharmaceutical giant is determined to protect both its intellectual property and patient safety in the face of mounting counterfeit threats. As authorities intensify enforcement and awareness efforts, American consumers are advised to ensure the authenticity of their medication—and investors are watching closely as the legal battles unfold.
